Courtesy of https://markcrispinmiller.substack.com
INDIA
Samajwadi Party’s Gorakhpur Lok Sabha candidate Kajal Nishad hospitalised due to cardiac arrest
April 8, 2024
Samajwadi Party candidate from Gorakhpur Lok Sabha constituency Kajal Nishad was rushed to a private hospital in Uttar Pradesh’s Lucknow after her health deteriorated on Sunday (April 7). Her husband said that she was experiencing some issues with her blood pressure and heart. Kajal was admitted to Star Hospital after her health She had suffered cardiac arrest. “We are taking her to Lucknow,” Kajal’s husband Sanjay Nishad told reporters. Kajal Nishad (41) is in the fray against actor and sitting BJP MP Ravi Kishan Shukla from the high-profile Gorakhpur seat. Kajal is a popular TV actress and has worked in various daily soaps, including Lapataganj.
MP Chandrasekhar Sahu hospitalized due to cardiac-related ailment
April 11, 2024
Berhampur MP Chandrasekhar Sahu [73] has been hospitalized following he complained of cardiac-related complications. Sahu was rushed to a private hospital in Berhampur in the morning for emergency medical intervention. Sahu’s health condition is reportedly stable. He is likely to be discharged after a few days said the doctor attending him.
SRI LANKA
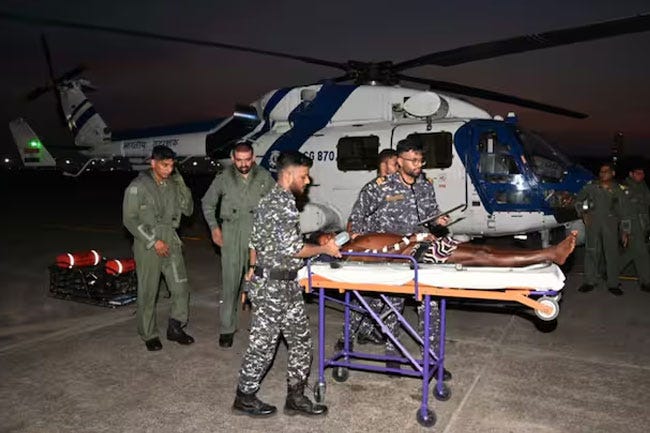
Indian Coast Guard rescues Sri Lankan fisherman after mid-sea medical emergency
April 6, 2024
The Indian Coast Guard evacuated a Sri Lankan fisherman facing a life-threatening medical emergency mid-sea on Friday. The Sri Lankan Fishing Vessel (SLFV) ‘Kalpeni’ ventured into the sea and it became adrift due to engine failure on April 1. One of the fishermen, aged 44, suddenly developed shortness of breath after the boat drifted 60 nautical miles from Chennai on April 5. Later, the fisherman was handed over to officials from Sri Lankan High Commission for further medical treatment.
SOUTH KOREA
Aespa’s Winter reportedly undergoes surgery for a collapsed lung
April 11, 2024
Aespa‘s Winter reportedly recently underwent surgery for a collapsed lung. According to an exclusive report from Tenasia, Winter [23] is currently recovering from surgery for pneumothorax (collapsed lung) at a hospital in Seoul. She underwent the surgery on April 11. Winter recently carried out her schedule in Japan prior to her surgery, appearing on the Japanese music show Venue 101. Pneumothorax surgery usually requires two to three days of hospitalization following the surgery and up to two months of recovery time for a full recovery. However, Aespa is scheduled to make their comeback in May and begin their world tour in June.
AUSTRALIA
Jimmy Barnes announces first NZ tour since heart surgery
April 4, 2024
Australian singer Jimmy Barnes has announced his first New Zealand tour since having open heart surgery in 2023. Barnes will play shows in Christchurch, Wellington, New Plymouth and Auckland in July, with his doctors giving him the all clear to return to performing. Barnes was hospitalised in November 2023 with bacterial pneumonia, however, he became very unwell, developing a staph infection which required surgery. The infection then progressed further causing endocarditis, a life-threatening condition that without treatment could have been fatal. This affected Barnes’ aortic valve and required urgent cardiac surgery to replace it.
No age reported.
Sasha has a disease so rare, it doesn’t have a name
March 26, 2024
Watching videos of their daughter’s birthday, David and Nadine Lipworth who lives in Australia can’t recognise the girl sitting on her father’s lap, singing happy birthday as she blows out four candles. Just months later, the mysterious genetic condition that had hampered Sasha’s development began whittling away at the small progress she’d made since birth. It robbed her of the ability to talk, play and complete basic tasks. The Lipworths celebrated her seventh birthday this month. She couldn’t blow out one candle. It’s unthinkable now that she was able to say those things and to do those things,” David Lipworth says.” Sydney researchers are beginning to piece together the molecular puzzle behind Sasha’s regression, in the hope of developing personalised treatments not only for her but for other children with rare and ultra-rare genetic conditions. After months of painstaking molecular detective work, University of Sydney Professor Sandra Cooper and her RNA for Rare Disease team this month identified the specific mutation in Sasha’s SLC6A1 gene that causes her condition. Clinical genetic testing found a suspicious variant in Sasha’s SLC6A1 gene, which was starving her of proteins that her brain needs.
NEW ZEALAND
Journalist Rachel Smalley’s family heartbreak: ‘In an instant, everything changed’
April 7, 2024
Auckland – The health journalist was blindsided when her mother was diagnosed with breast cancer. Last November 16, Rachel Smalley was on top of the world. The former broadcaster had just taken a call from TVNZ Sunday journalist Miriama Kamo to tell her she’d been nominated for the Kiwibank New Zealander of the Year 2024 awards, and she couldn’t wait to tell her mum Shirley Mackie. “I was so excited – it was a huge surprise,” says Rachel, 52, who was nominated for her work with The Medicine Gap, an initiative advocating for better access to modern medicine. “Miri told me I had to keep the news quiet, but I’d been given the okay to tell Mum. I celebrated for about 90 seconds, got quite emotional about it – and then I called Mum with the news. But her voice just broke. I thought maybe she was getting emotional about my news, but she told me she had been diagnosed with breast cancer that very morning. In an instant, everything changed.” Two weeks earlier, Shirley, 76, had booked a private mammogram, after a previous conversation with Rachel about getting one. When Shirley made the appointment, the nurse asked if she had any problems. “I didn’t,” she says. “I’m very aware of my body and I check my breasts.” But about a week later, before the mammogram, she looked in the mirror. “I realised I had a lump about the size of half a marshmallow, high on the side of my right breast. It was soft to the touch, but when I put pressure on it, it hurt.”
Six-year-old with terminal cancer finds joy in his 100 birds: ‘The king of the birds’
April 7, 2024
West Auckland – Known lovingly amongst friends as “the bird man”, 6-year-old Caiden MacGibbon got his first pet bird shortly after being diagnosed with an incurable brain stem tumour in 2022.Now, 16 months on, walking into the family’s West Auckland home means walking into the sound of more than 100 feathered creatures singing – scattered between indoor cages and an outdoor aviary. It was November 2022 that Caiden was diagnosed with an untreatable cancer – grade 4 DIPG (diffuse intrinsic pontine glioma). It is an extremely aggressive tumour of the brain-stem, which mainly affects children. He’s had two rounds of radiation therapy and the family “don’t know” what the next step will be, but for now, Jasmine and Karl MacGibbon are focussed on making Caiden happy.
Temuka family fundraises more than $8.5k in House to House
March 30, 2024
Temuka, Canterbury – When 4-year-old Grace Quinlan became sick last August her family took her to the doctor where blood tests and X-rays did not show anything out of the ordinary. But two months later, and still unwell, the Temuka girl was referred to a paediatrician in Timaru, her mother Kaye Duncan said. “The paediatrician saw Grace and did a follow up blood test and saw that levels had not returned to normal,’’ Duncan said. The family was then referred to the Child Haematology and Oncology Centre in Christchurch and within a week Grace was diagnosed with leukaemia. The diagnosis came as a “big shock’’ to the family as they had hoped Grace had the flu or Covid-19 as she had showed signs of improvement before seeing the paediatrician. Duncan’s advice to other parents was to “follow their instincts” if they felt something was wrong with their child.
Dunedin mum shares cancer battle
April 7, 2024
Dunedin, Otago – For the past year, Dunedin mum Tamsyn Hayes has been battling an aggressive form of breast cancer, while striving to maintain family life and her career as a policewoman. Ms Hayes is undergoing treatment for stage 3, grade 3 triple-negative breast cancer and is keen to highlight the fact cancer can strike anyone at any age. The former multiple ironman and triathlon competitor said it was “easy to think that cancer doesn’t happen to fit, young healthy people, but it does“.
No age reported.
Click this link for the original source of this article.
Author: Mark Crispin Miller
This content is courtesy of, and owned and copyrighted by, https://markcrispinmiller.substack.com and its author. This content is made available by use of the public RSS feed offered by the host site and is used for educational purposes only. If you are the author or represent the host site and would like this content removed now and in the future, please contact USSANews.com using the email address in the Contact page found in the website menu.